Project Consulting Services
Custom Assay Development
Method Transfer to QC or Third-Party
ELISA Kit Development
Reporter Cell Line Development
Release Assay Development
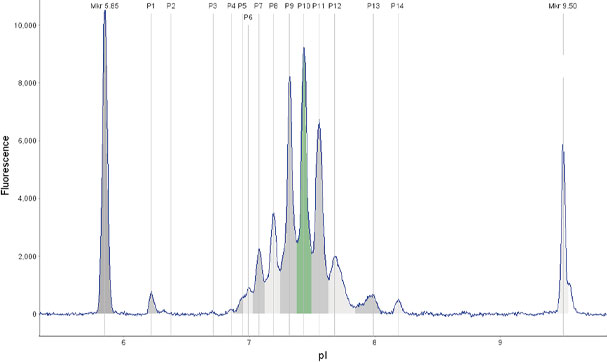
icIEF分析双特异性抗体的电荷变异体
In-silico
Analysis
LC-MS
MDR
Method
Pre-validation
Method Development
Report (MDR)






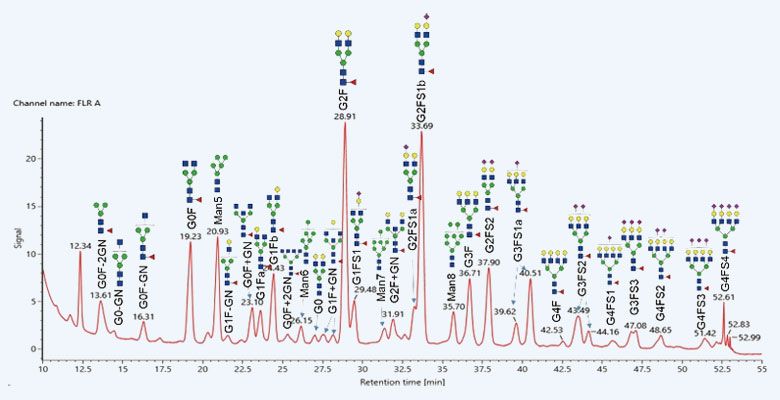
复杂游离N-糖谱鉴定图谱(HILIC-FLR-MS)
Sequence Coverage
Post-Translational Modifications (PTMs)
N-/C-Terminal Sequence Confirmation
Sequence Variant Analysis
Cleavage Site Analysis
Site-Specific Glycosylation Analysis
Techniques Include:
Circular Dichroism (Far-UV/Near-UV CD)
Differential Scanning Calorimetry (DSC)
Fluorescence Spectroscopy
Size-Exclusion Chromatography with Multi-Angle Light
Scattering (SEC-MALS)
Analytical Ultracentrifugation (AUC)
Dynamic Light Scattering (DLS)
Micro-Flow Imaging (MFI)

Project Consulting Services
Custom Assay Development
Method Transfer to QC or Third-Party
ELISA Kit Development
Reporter Cell Line Development
Release Assay Development

Comprehensive ELISA Analysis
SPR/BLI Affinity Analysis
In Vitro Bioactivity Analysis
Multicolor Flow Cytometry Analysis
Early-Stage Molecular Screening
Exosome Analysis
ELISA binding potency
ELISA competitive binding
ELISA titer
ELISA kit construction
Reporter gene assay
Cell cytotoxicity
Cell proliferation
ADCC/ADCP/CDC
Cytokine release
Enzymatic activity
Virus neutralization
SPR/BLI
Affinity screening
Epitope screening
ddPCR/qPCR) c/gDNA analysis
Mycoplasma
Host cell protein
Host cell DNA
Protein A
FVIISelect
Insulin
Ampicillin
Dnase/RNase
Coagulation Activity
Fibrinolytic Activity
Western Blot
Endotoxin

Design reporter cellline constructionplan based on theproduct MOA andclient requirements.

Construct high-efficiency expressionvectors containing thetarget gene andreportergene.

Chemical
Electroporation
Lentiviral

Confirm gene expression
Validate reporter genefunction
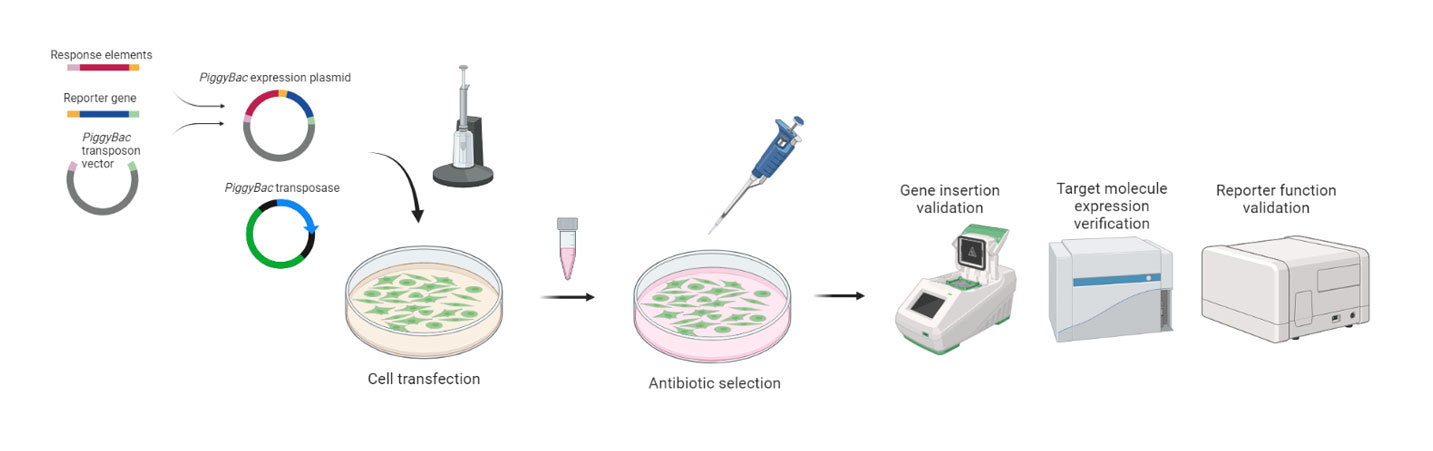
3-5 Weeks
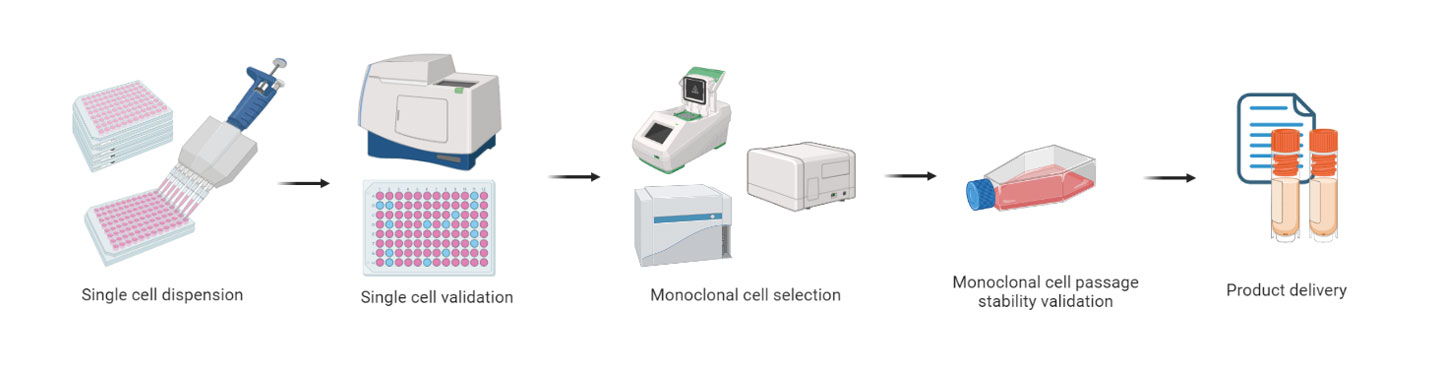
Single-cell seeding
Monoclonal gene expression verification
Monoclonal functional validation
7-8 Weeks
Functional testing ofmonoclonal cells atdifferent passages
Gene expression testing ofmonoclonal cells atdifferent passages
1 Week
Stable monoclonal cell line
Product quality test report(CoA)
Cell line construction report





|
Cell Type |
Product Name |
Application |
| ADCC & ADCP | ADCC Reporter Jurkat (158V) |
Detection of Antibody ADCC Effect |
| ADCP Reporter Jurkat (131H) |
Detection of Antibody ADCP Effect |
|
|
Target and Pathway Research |
NF-κB Reporter 293T |
Development of NF-κB Pathway Activators or Inhibitors |
| TLR4 Reporter 293T |
Screening of Functional TLR4 Antibodies / Ligands |
|
| STAT3 Reporter DH82 |
Development and Screening of STAT3 Pathway Activators or Inhibitors |
|
|
Viral Drug Research |
Universal Virus-inducible Reporter Cell |
Detection of Antiviral Drug Activity |
|
Overexpressing Cells |
Canine IL-31R overexpressing 293T |
Development and Screening of Canine-Derived IL-31 Receptor Antibodies or Canine-Derived IL-31 Blocking Antibodies |
|
Category |
Testing / Method Development Item |
Analytical Method |
|
Protein Content |
UV280/SoloVPE | |
| BCA/Bradford/SDS-PAGE | ||
| RP-HLPC/SEC-HPLC/Protein A | ||
|
Identity |
Isoelectric Point |
icIEF |
|
Peptide Mapping |
RP-HPLC/UPLC | |
|
Western Blot |
Western Blot | |
|
Biological Activity |
Binding Activity |
ELISA |
|
Cell-Based Activity |
MoA-based | |
|
Primary Structure |
Mass Spectrometry Molecular Weight
|
Intact Mass |
|
Reduced Mass |
||
|
Deglycosylated Intact Mass |
||
|
Deglycosylated Reduced Mass |
||
|
Sequence Coverage |
Multi-Enzymatic LC-MS Peptide Mapping |
|
|
Post-Translational Modifications |
LC-MS Peptide Mapping |
|
|
N-Terminal / C-Terminal Sequence Analysis |
LC-MS Peptide Mapping |
|

